Interim Order Respecting the Importation, Sale and Advertising of Drugs for Use in Relation to COVID-19
I have concerns — the Government of Canada has quietly allowed something that on its face is benign, but in reality is anything but. They have granted the ability for foreign regulators to be able to export their COVID drugs and vaccines into Canada for phase two and three trial testing on Canadians. Phase two and three trials can contain anywhere from a couple hundred, to three thousand people.
Now bear in mind, the government ran this discussion hidden from the public yesterday. They held the discussion on teleconference, only available to journalist who had both the number, and the passcode to get into the conference. This was allegedly done to help quell Canadians fears about this exact move — something that could’ve been more easily achieved by actually talking to Canadians, not journalists.
As I was on the call, I was frantically trying to research what was being discussed before the question period arose. They mentioned the interim order, and until that point I had not been aware that this document existed. As I combed through the document, I found something that left me uneasy.
“Instead of providing detailed reports of the tests establishing the safety of a new drug and substantial evidence of clinical effectiveness as required by the Food and Drug Regulations, the Interim Order requires an applicant to submit the known information with respect to the safety and effectiveness of a COVID-19 drug.”
What is the known information that an applicant is required to submit in regards to the safety and effectiveness of a COVID-19 drug or vaccine?
“In order for a drug to be eligible to use the expedited pathway for drugs that are authorized by a foreign regulatory authority, the drug must be included on the List of Foreign Drugs, which is maintained by the Minister and incorporated by reference in the Interim Order. A drug may be included on this list if it has been shown to provide benefit in the context of the COVID-19 pandemic, and it has received an authorization for sale in a foreign jurisdiction.”
What does this mean?
Let me get this straight the “drug may be included on this list if it has been shown to provide benefit in the context of the COVID-19 pandemic, and it has received an authorization for sale in a foreign jurisdiction?” This means that if China gives approval for a drug in China, and they show it to “provide benefit in the context of the COVID-19 pandemic” it very well could end up being tested here in Canada.
Safeguards
“The applicant must submit evidence that the drug is authorized for sale in a foreign jurisdiction and sign an attestation that, if requested, all of the information used to authorize the drug by the foreign regulatory authority will be made available to the Minister.”
Isn’t it comforting? — To know that the government doesn’t require that all information be made available to the minister before its potential use in Canada. Admittedly, they have to sign an attestation that if requested they will give all of the information used to authorize the drug — by why not demand it from the beginning?
Not all regulators are created the same
What nations regulators do you think Canadians would prefer to trial drugs and vaccines from? If given the choice would you desire a vaccine that is being trialed on Canadians to potentially come from China? I know that I wouldn’t want any Chinese vaccine to be trialed on Canadians, and the reason is rather simple — lack of transparency and trust.
Chinese Alzheimer drug approval
The distrust I have for approved vaccines and drugs out of China being tested on Canadians comes from a very legitimate place — a previous history of falsifying studies to approve drugs. One Chinese company received approval to use their Alzheimer’s vaccine, but many of the studies surrounding the approval come with great scrutiny — and not a lot of transparency.
“One notable feature is the steep drop in the scores of the placebo group between week 24 and week 36, which is rather strange, and for which no explanation is apparently given. And if I’m interpreting this correctly, both the placebo and treatment groups improved at the earlier time points, it’s just that the treatment group improved more?”
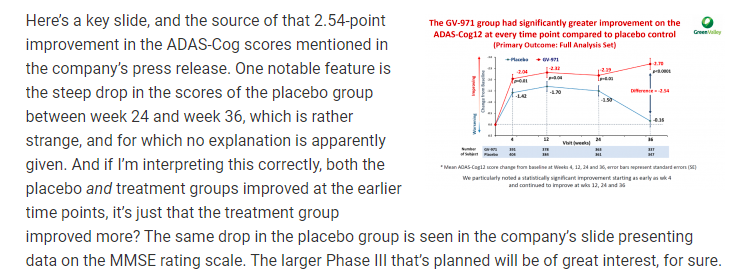
So let me get this straight, not only was there a steep drop off in the placebo group, not only did the placebo and non-placebo groups improve together in unison, not only is the phase two data not available, but the placebo group falls off the face of the planet and no explanation is given? This type of fine regulatory approval could allow for drugs and vaccines to make their way to Canada to be tested on Canadian’s for COVID.
This interim order could mean that Canadians would receive questionable drugs and vaccines from foreign regulators that aren’t approved by Health Canada, and these could be tested on up to 3000 individuals at a time. Mind you, the people who are taking these trial vaccines and drugs know there are risk associated — but they also likely believe that the studies that allowed them to be trialed in the first place should pass the sniff test.
So what kind of regulators could be included? For starters the W.H.O., and China’s SFDA could be considered – but take a look at the list yourself.
Accelerated vaccine development
Vice has a video that discusses the typical timeline for vaccine development, and how those racing to create a vaccine are seeking to condense this timeframe dramatically (average 10 years into 1.5 years). They also point to over 30,000 participants who have signed up to do what is known as a human challenge trial.
A challenge trial is where the subjects involved in the study are intentionally exposed to the condition that is being tested for — so in this example, exposing the subjects directly to COVID-19. Many of those Vox interviewed who agreed to participate in a human challenge trial seemed to do so out of a desire to help others.
It also stated in the video that all except one vaccine study has refused to participate in human challenge trial, deeming it “unethical” to participate in a human challenge trial. The one institution that is considering a human challenge trial is the developers at Oxford University who have stated that they hope to have one of these trials ready by the end of the year.

Who funds the W.H.O?
Many also have concerns about the World Health Organization (WHO) being compromised due to the large amount of funding they receive from Bill and Melinda Gates various foundations. “However, in terms of brass tacks, the second largest voluntary contributor to the W.H.O is the Bill & Melinda Gates Foundation, which pays about $531 million.” Bill Gates organization Gavi is the third largest contributor to W.H.O, donating ” $370 million to the WHO, with about 72.36 percent of its funding going toward vaccines and preventable disease.”

Canadians must demand answers from their government on this interim order. We must ask if our government would take a vaccine or drug from the W.H.O, or China to test on Canadians in phase two and three trials (potentially thousands at a time). The answer, as is evident from Trudeau’s wiliness to take a vaccine from China earlier in the “pandemic” is likely yes.
Let’s demand that we don’t take a vaccine from China – we can’t trust their studies, no matter how promising they “look”. The W.H.O has proven that their expertise are anything but, and have proven themselves an unreliable source of information during these times at best.
@DivergeMediaNews via Facebook
If you like our work, and want to support independent Canadian media that covers the stories that matter, please consider donating in the form below. We will have merch to offer soon!
Make a one-time donation
Make a monthly donation
Choose an amount
Or enter a custom amount
Your contribution is appreciated.
Your contribution is appreciated.
DonateDonate monthly
